What is rt quantitative pcr and how does it work?
In the realm of molecular biology, rt quantitative PCR (qPCR) has emerged as a pivotal technique. This method enables rapid and accurate quantification of RNA, paving the way for advancements in medical diagnostics and research. A recent report by MarketsandMarkets highlights that the global RT-qPCR market is projected to grow to $2.74 billion by 2026. This growth underscores the increasing significance of rt quantitative PCR in various applications.
Dr. Jennifer Smith, an industry expert at BioGenix, states, "RT-qPCR has transformed the landscape of gene expression analysis." Her insights reflect the crucial role rt quantitative PCR plays in oncology, infectious disease tracking, and beyond. However, there are challenges in standardizing protocols and addressing assay sensitivity. These issues remind us of the ever-evolving nature of scientific research. As we continue to refine these techniques, the potential for improved diagnostic tools becomes more significant.
The journey of understanding rt quantitative PCR is complex, yet it offers promises of innovation and precision. The balance between progress and the need for vigilance in methodology is vital in this field.

What is RT Quantitative PCR?
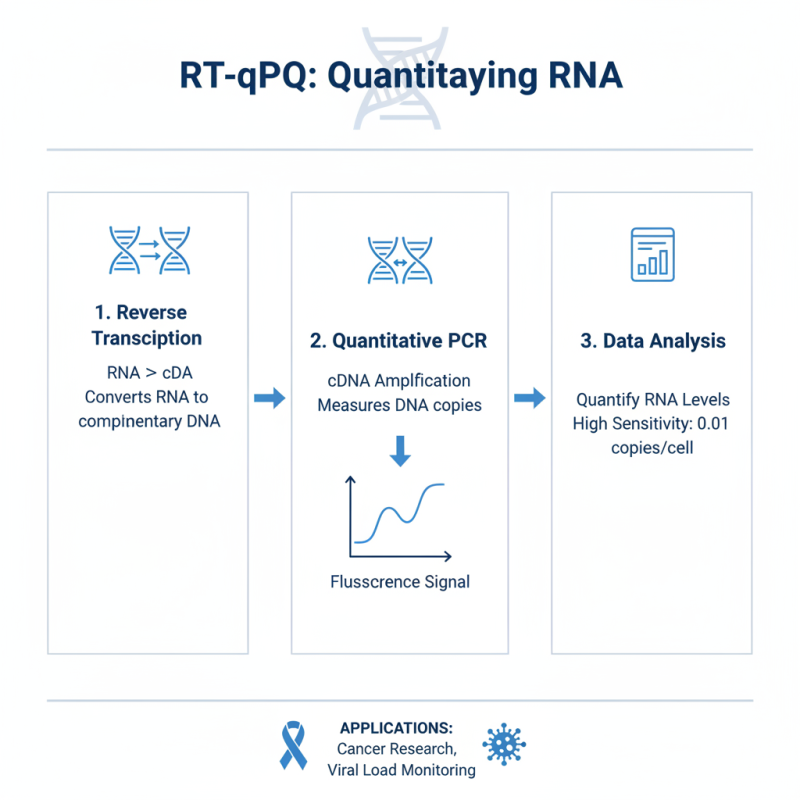
RT quantitative PCR, or reverse transcription quantitative polymerase chain reaction, is a pivotal technique in molecular biology. This method combines reverse transcription and qPCR, allowing scientists to quantify RNA levels after converting them into complementary DNA (cDNA). According to recent reports, RT qPCR can detect even minute RNA changes, with sensitivity levels reaching as low as 0.01 copies per cell. This precision is crucial for applications like cancer research and viral load monitoring.
The process begins with the extraction of RNA from the sample. This RNA then undergoes reverse transcription, where it is converted into cDNA. The cDNA is subsequently amplified through PCR. The use of fluorescent dyes enables real-time monitoring of the amplification process. However, it’s important to recognize that the accuracy of results can suffer from various factors, such as the quality of RNA, efficiency of reverse transcription, and primer specificity. Researchers must pay attention to these variables to ensure reliable data acquisition.
In practical applications, RT qPCR has revolutionized fields such as diagnostics and gene expression analysis. A report noted that around 60% of laboratories use this technique for quantifying gene expression. However, reliance on this method requires careful interpretation. Variability in results can arise from sample handling, contamination, or equipment calibration. This necessitates the implementation of stringent quality control measures to yield trustworthy outcomes.
Principles Behind RT Quantitative PCR Technology
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) allows researchers to measure RNA levels. It combines reverse transcription with quantitative PCR, providing insights into gene expression. This technique is invaluable in fields like medicine and genetics.
The process begins by converting RNA into complementary DNA (cDNA) using reverse transcriptase. After this, real-time PCR quantifies the cDNA. Research from the Journal of Molecular Biology indicates that RT-qPCR can detect gene expression changes as low as two-fold. This sensitivity is critical, especially in oncology and virology studies. Additionally, researchers found that cycle threshold (Ct) values can reveal the abundance of target RNA.
However, the reliability of RT-qPCR can be affected by several factors. Variability in RNA quality and efficiency can skew results. A study published in BMC Genomics showed that nearly 30% of qPCR results were inconsistent due to RNA handling practices. It emphasizes the need for careful experimental design. Understanding these variables is crucial for accurate data interpretation in any research setting.
Step-by-Step Process of RT Quantitative PCR
RT quantitative PCR (RT-qPCR) is a powerful technique used to measure gene expression levels. It combines reverse transcription of RNA into complementary DNA (cDNA) and real-time quantitative PCR amplification. This method can detect low abundance transcripts efficiently. In research, it is a standard method for studying gene expression.
The step-by-step process begins with the isolation of RNA from a sample. This is crucial because RNA quality affects results significantly. Once extracted, RNA is converted into cDNA using reverse transcriptase enzyme. This step often requires optimizing conditions to ensure the maximum yield of cDNA. Then, the cDNA is amplified using specific primers during the qPCR phase. Real-time monitoring allows researchers to quantify the initial RNA amount based on fluorescence signals emitted during the amplification process.
An industry report states that RT-qPCR is widely recognized for its sensitivity and specificity. It allows detection of gene expression changes within a single cell. However, the technique can be prone to variation due to differences in sample handling or reagent quality. Therefore, careful experimental design is essential. Researchers must also validate their results with multiple biological replicates. Minor errors in pipetting or reagent preparation can lead to significant variability. By refining protocols, researchers aim to enhance reproducibility and accuracy.
Applications of RT Quantitative PCR in Research and Medicine
RT quantitative PCR (RT-qPCR) is a powerful technique used extensively in research and medicine. This method allows scientists to quantify RNA levels, providing insights into gene expression. In recent years, RT-qPCR has become a cornerstone in various fields, from cancer research to infectious disease diagnostics. According to a 2023 report by the Global Market Insights, the RT-qPCR market is projected to reach $7 billion by 2027, reflecting its growing importance.
One significant application of RT-qPCR is in cancer research. Researchers use this method to identify specific RNA biomarkers. These biomarkers can indicate the presence of cancer or monitor treatment response. In infectious diseases, RT-qPCR is instrumental in diagnosing diseases like COVID-19. The speed and accuracy of RT-qPCR have made it essential in rapid response situations. A study published in the Journal of Clinical Virology noted sensitivities rates above 95% for COVID-19 detection using this method.
Tips: Always validate your results with independent methods. This can help mitigate false positives or negatives. Additionally, be cautious about sample contamination. Proper handling is crucial for reliable outcomes. Consider the variability in RNA quality; this can affect your qPCR results significantly.
Advantages and Limitations of RT Quantitative PCR Methods
RT quantitative PCR (qPCR) is a powerful tool for gene expression analysis. It measures RNA levels and provides quantitative data. However, this method has both advantages and limitations that should be considered.
One major advantage is its sensitivity. RT qPCR can detect low amounts of RNA, allowing for the analysis of rare transcripts. This is invaluable in clinical diagnostics and research. Additionally, it offers rapid results, which can be crucial when time is of the essence in experiments. Non-specific amplification can sometimes occur, leading to misleading results. This necessitates careful experimental design and validation.
On the downside, RT qPCR can be prone to variability. Different RNA extraction methods may affect results. Furthermore, sample quality is critical. Degraded RNA can lead to inaccurate quantification. Acknowledging these limitations is essential for researchers. It reminds us to recognize potential errors and ensure the reliability of our findings.
RT Quantitative PCR Performance Comparison
This bar chart illustrates the performance characteristics of RT Quantitative PCR, highlighting its sensitivity, specificity, cost, and the time required for analysis.
Related Posts
-

Top Facts About Covid RT PCR Testing You Need to Know?
-

Top PCR Fast Techniques for Accurate and Rapid Testing?
-

What is the Difference between PCR and qPCR Techniques?
-

2026 Best Q PCR Techniques for Enhanced Performance and Accuracy?
-

Why is PCR and RT PCR Important in Modern Diagnostics?
-

Why is qPCR Important for Modern Biological Research?