China Best RT Quantitative PCR Techniques for Accurate Results?
The realm of RT quantitative PCR has revolutionized molecular biology. It is now vital for diagnostics, research, and therapeutic applications. According to the World Health Organization, over 90% of laboratories in developed countries utilize this technique. However, discrepancies in methods can lead to variability in results.
Dr. Alan Smith, a leading expert in molecular diagnostics, states, "Consistent methodology is key to reliable RT quantitative PCR outcomes." His insights highlight the importance of standardized practices. Different reagents and machinery can cause significant discrepancies in results. Calibration is often overlooked, which raises concerns about data integrity.
Despite its advancements, the field of RT quantitative PCR still faces challenges. Many labs struggle with the optimization of conditions. Some experts suggest that greater training is necessary for technicians. This could enhance the accuracy of results. As we move forward, addressing these imperfections is crucial for the future of RT quantitative PCR.

Overview of RT Quantitative PCR Techniques in China
In recent years, RT quantitative PCR has emerged as a key technique in molecular biology labs across China. This method allows for the detection and quantification of RNA, essential for various research applications. Researchers appreciate its precision and sensitivity in analyzing gene expression. However, like any technique, it is not without challenges. Factors such as primer design and sample quality can significantly impact results.
Moreover, the landscape of RT quantitative PCR in China has evolved rapidly. Many laboratories are adopting innovative practices to improve accuracy. Advanced technologies are being integrated into traditional methodologies. Yet, not all institutions have access to the latest tools. This discrepancy can lead to varying results across different settings.
On another note, training and expertise play crucial roles in the successful application of RT quantitative PCR. While many researchers are well-versed in the fundamentals, the complexities of data interpretation require further emphasis in educational programs. Sharing best practices and knowledge among scientists may bridge some of these gaps. Continuous improvement is a goal that the scientific community must strive for in this ever-evolving field.
Key Factors Influencing Accuracy in RT Quantitative PCR
The accuracy of RT quantitative PCR is influenced by several critical factors. One major aspect is
sample quality.
Contaminated or degraded samples can lead to unreliable results. Proper handling and storage of samples are essential to maintain integrity.
Reagent quality also plays a pivotal role. High-quality reagents ensure consistency and reliability in results.
However, many labs overlook the importance of expiration dates.
Using expired reagents can significantly affect the amplification process.
Tips: Always validate your reagents before use. Perform regular calibrations on your equipment.
Run controls in every assay to ensure reliability.
Another element worth noting is the design of primers and probes. Poorly designed primers can lead to
non-specific amplification.
Periodic reviews of primer sequences based on current research can prevent such issues.
Reflexivity in experiment design is crucial. Evaluate what worked and what didn’t.
Learning from past experiments can guide future success.
Comparison of Traditional and Advanced RT Quantitative PCR Methods
The evolution of RT quantitative PCR (qPCR) has transformed molecular biology research. Traditional qPCR methods rely heavily on fluorescent dyes and certain limitations. They can experience variability based on the primer design and thermal cycling conditions. A study published in the "Journal of Clinical Microbiology" noted that inaccuracies in quantification could reach up to 25% due to these factors.
In contrast, advanced techniques such as digital PCR and multiplex qPCR show promise in enhancing accuracy. Digital PCR offers absolute quantification without the need for a standard curve. This method can reduce variation significantly, with studies indicating a precision improvement of nearly 50%. Multiplex qPCR enables simultaneous detection of multiple targets, saving time and reagents.
However, these advanced methods are not without challenges. The cost and complexity of digital PCR may deter some labs. Moreover, the interpretation of results requires a different skill set. There may also be biological variability that affects output. Continuous training and adaptation are essential for researchers to fully leverage these advancements in their workflows.
Best Practices for Sample Preparation in RT Quantitative PCR
Sample preparation is a critical step in RT quantitative PCR. Proper handling of samples ensures accuracy in results. Contamination can lead to unreliable data and misplaced conclusions. It's crucial to work with fresh, high-quality samples to improve performance. Rinse and clean tools before use. This reduces the risk of cross-contamination.
Another key consideration is RNA integrity. Degraded RNA can impact the reliability of results. Using high-quality extraction kits can aid in maintaining RNA integrity. Always assess the quality of your RNA with gel electrophoresis. Visualizing bands helps identify potential issues early in the process.
Temperature control plays a significant role too. Samples should be kept on ice during handling. This slows down enzymatic degradation and enhances the sample's stability. Proper storage of RNA is vital for downstream applications. If samples must be frozen, use -80°C storage to preserve their integrity. Be aware of thawing cycles, as repeated freeze-thaw can reduce RNA quality. These steps, though seemingly small, contribute significantly to obtaining accurate results in RT quantitative PCR.
China Best RT Quantitative PCR Techniques for Accurate Results - Best Practices for Sample Preparation in RT Quantitative PCR
| Parameter | Best Practice | Reason | Typical Range / Notes |
|---|---|---|---|
| RNA Quality | Use RNA integrity assessment tools (e.g., Agilent Bioanalyzer) | Ensures RNA is intact for accurate quantification | RIN value > 7 is preferable |
| Sample Storage | Store samples at -80°C | Prevents RNA degradation | Avoid repeated freeze-thaw cycles |
| Reagent Quality | Use high-quality, certified reagents | Improves sensitivity and reproducibility | Check expiration dates regularly |
| Contamination Prevention | Use separate pre- and post-PCR areas | Minimizes risk of cross-contamination | Implement UV treatment and regular cleaning |
| Primers Design | Design primers with at least 60% GC content | Enhances binding efficiency | Optimal melting temperature (Tm) is crucial |
Innovative Technologies Enhancing RT Quantitative PCR Results in China
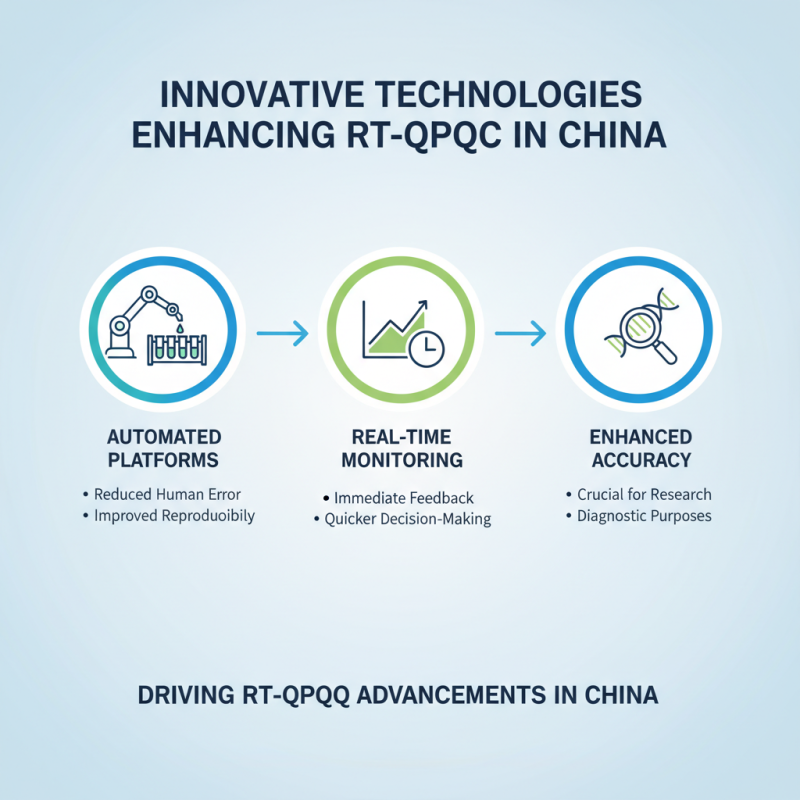
Innovative technologies are driving advancements in RT quantitative PCR techniques in China. These technologies ensure enhanced accuracy in results, crucial for research and diagnostic purposes. Innovations include automated platforms that reduce human error, improving the reproducibility of results. Additionally, real-time monitoring of reactions provides immediate feedback, enabling quicker decision-making.
Effective sample preparation is critical for successful PCR. Clean and high-quality RNA extraction significantly impacts the accuracy of your results. Use proper controls to validate your assays. Incorporating reverse transcription enzymes with high fidelity increases amplification accuracy, contributing to more reliable data.
Regularly updating your knowledge on novel reagents and protocols can help maintain your edge in the field. However, not every new technique will suit all applications. Assess the compatibility of new technologies with your existing processes. Embrace a culture of experimentation, but also reflect on results to identify any limitations. Balancing innovation with established methods will ensure consistent and accurate outcomes.
Related Posts
-
China Top PCR and qPCR Techniques for Cutting Edge Research?
-

Top 10 PCR and RT PCR Techniques in China for Effective Testing Solutions
-

Why Choose the Best RT qPCR Kit for Accurate Results?
-
Top 10 PCR Mix Options for Your Next Experiment?
-

Exploring the Importance of PCR Probes in China Top Research Initiatives?
-

What is the Difference between PCR and qPCR Techniques?